Introduction
Each physical quantity is measured against a randomly selected and internationally recognized reference standard, known as a unit. The result of measuring a physical quantity is expressed by a number (or numeric scale) together with a unit. Although the number of physical quantities seems very large, we only need a limited number of units to express all physical quantities because they are related to each other. Base units or base quantities are referred to as base units. Units of other physical quantities can be expressed as a set of base units. These units obtained for derived quantities are called derived units. The complete set of these units, both base units, and derived units is called the system of units.
The International System of Units
In earlier times scientists of different countries were using different systems of units for measurement. Three such systems, the CGS, the FPS (or British) system, and the MKS system were in use extensively till recently.
The widely accepted standard units for length, mass, and time in these systems were as follows:
- CGS: where length was measured in centimeter, mass in gram and time in second.
- FPS: where length was measured in foot, mass in pound and time in second.
- MKS: where length was measured in meter, mass in kilogram and time in second.
The system of units which is currently accepted all over the world for measurement is the Système Internationale d' Unittes. (French for international system of units), abbreviated as SI. The SI, with standard scheme of symbols, units and abbreviations, developed by the beureau International des Poids et measures (The International bureau of Weight and Measures).
In SI unit there are seven fundamental units which are given below in the table.
| Base Quantity | Name | Symbol | Definition |
|---|---|---|---|
| Length | meter | m | The meter is the SI unit of length. It is defined by taking the fixed numerical of the speed of the light in vacuum to be 299792458 when expressed in the term of m/s, where second is defined in term of the caesium frequency ∆νcs. |
| Mass | kilogram | kg | It is defined by taking the fixed numerical value of the Planck constant h to be 6.62607015×10–34 when expressed in the unit J s, which is equal to kg m2 s–1, where the meter and the second are defined in terms of c and ∆νcs. |
| Time | second | s | One second is defined by taking the fixed numerical value of the caesium frequency∆νcs, the unperturbed ground-state hyperfine transition frequency of the caesium-133 atom, to be 9192631770 when expressed in the unit Hz, which is equal to s–1. |
| Electric Current | ampere | A | One ampere is defined by taking the fixed numerical value of the elementary charge e to be 1.602176634×10–19 when expressed in the unit C, which is equal to A s, where the second is defined in terms of ∆νcs. |
| Temperature | kelvin | K | One kelvin is defined by taking the fixed numerical value of the Boltzmann constant Temperature k to be 1.380649×10–23 when expressed in the unit J K–1, which is equal to kg m2 s–2 k–1, where the kilogram, meter and second are defined in terms of h, c and ∆νcs. |
| Amount of substance | mole | mol | One mole substance contains exactly 6.02214076×1023 elementary entities. This number is the fixed numerical value of the Avogadro constant,NA, when expressed in the unit mol–1 and is called the Avogadro number. The amount of substance, symbol n, of a system is a measure of the number of specified elementary entities. An elementary entity may be an atom, a molecule, an ion, an electron, any other particle or specified group of particles. |
| Luminious intensity | Candela | cd | The candela, symbol cd, is the SI unit of luminous intensity in given direction. It is defined by taking the fixed numerical value of the luminous efficacy of monochromatic radiation of frequency 540×1012 Hz, Kcd, to be 683 when expressed in the unit lm W–1, which is equal to cd sr W –1, or cd sr kg–1m–2s3, where the kilogram, metre and second are defined in terms of h, c and ∆νcs. |
In addition to the seven basic units, there are two other units. The units define for:
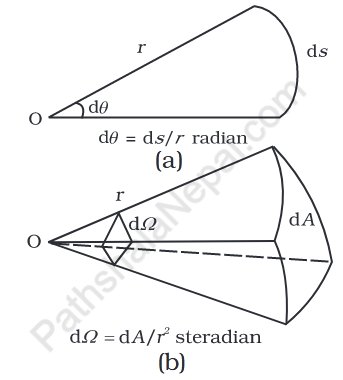
- the plane angle dθ as the ratio of the arc length ds to the radius r and
- the solid angle dΩ as the ratio of the cross-sectional area dA to the surface of the sphere described for vertex O as the center.
To the square of radius r as shown in the figure. 2.1 (a) and (b). The unit of the angle is the radian with the symbol rad and the unit of the solid angle is the steradian with the symbol sr. Both quantities are dimensionless.
Measurement of Length
The standard unit of length is meter. But there are also units of measurement of length when we need to measure either very small distances or very large distances. For example, a meter scale is used for lengths from 10–3 m to 102 m. A vernier calipers is used for lengths to an accuracy of 10 –4 m. A screw gauge and a spherometer can be used to measure lengths as less as to 10–5 m. To measure lengths beyond these ranges, we make use of some special indirect methods.
Measurement of Large Distances
Distances as big as a planet or a star from Earth that cannot be directly measured by a meter is measured with the principle of parallax. When you hold a pencil in front of you against some specific point on the background (a wall) and look at the pencil first through your left eye A (closing the right eye) and then look at the pencil through your right eye B(closing the left eye), you would notice that the position of the pencil seems to change with respect to the point on the wall. This is called parallax. The distance between the two points of observation is called the basis. In this example, the basis is the distance between the eyes.
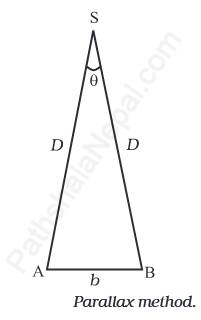
To measure the distance D of a distant planet S using the parallax method, we observe it from two different locations (observatory) A and B on Earth, simultaneously separated by a distance AB = b, as shown in the figure below. We measure the angle between the two directions in which the planet appears at these two points. The ASB in Figure below, denoted by the symbol θ, is called the parallax angle or parallactic angle.
As the planet is very far away, \(\frac bD<<1\), and therefore, θ is very small. Then we can approximately take AB as an arc of length b of a circle with center at S and the distance D as the radius AS = BS so that AB = b = D θ where θ is in radians.
\(D=\frac b\theta\)
Once we find D, we can similarly determine the size or the angular diameter of the planet. If d is the diameter of the planet and α the angular size (the angle subtended by d at the earth), we have α = d / D
Estimation of Very Small Distances (Size of a Molecule)
To measure small sizes, such as that of a molecule (10–8 m to 10–10 m), a special approach must be adopted. We cannot use a screwdriver or similar application. Even the microscope has some limitations. The microscope uses visible light to look at the study system. Finest example of measuring smaller distance is the the measurement of molecular size of an Oleic acid. In fact, the molecular size of Oleic acid is in the larger range compared to other molecules. Due to the special property of Oleic acid, Oleic acid when dissolved in water its molecules will stand up and support one another on end because of the attractive forces between the hydrophobic parts of the molecules. It like putting oil or vegetable ghee in water.
Here is how we measure the molecular size of an Oleic acid.
If ‘v’ is the volume of one drop, then the volume of n drops of the solution is ‘nv’.
Volume of oleic acid in n drops of solution = \(\frac{nv}{400}\) cc
If A cm2 is the area and ‘t’ its thickness then assuming the thickness is equal to one layer of molecules we can calculate the thickness
\(A\times t=\frac{nv}{400}\)
This means that the thickness is
\(Thickness=\frac{Volume}{Area}\)
\(t=\left(\frac{nv}{A\times400}\right)\)
Based on the assumption that the layer is one molecule thick, we obtain the value of the size of the Oleic acid molecule. The thickness value comes out to be in the order of 10-9 m. Considering how simple this demonstration is, this method has very good accuracy.
Range of Lengths
Objects and their distance comes in extremely wide range. From the smallest sub atomic particles to the far distant galaxies, it is very cumbersome to measure distances with one scale. For example, the size of a nucleus is in the order of 10-14 m while the size of observable universe is in the order of 1026 m.
We can use certain special length units for short and large length. These are
| Special Length Unit | Symbol | Meter Equivalent |
|---|---|---|
| 1 Fermi | f | 10-15 m |
| 1 angstrom | Å | 10-10 m |
| 1 Astronomical unit | AU (average distance from sun to earth) | 1.496×1011 m |
| 1 light year | ly | 9.46×1015 m |
| 1 Parsec | Parsec (distance at which average radius of earth's orbit subtends an angle of 1 arc second) | 3.08×1016m |
Measurement of Mass
A fundamental property of any object in the universe is its mass. Probably light particle (photon) is the only one object in nature which has no mass. Mass is independent of any other physical quantities like pressure, temperature or position in gravitational or electromagnetic field. The SI unit of mass is
kilogram (kg). It is defined by taking the fixed numerical value of the Plank Constant h to be 6.62607015×10–34 when expressed in the unit of Js which is equal to kg m2s–1, where the meter and the second are defined is terms of C and ∆νcs
While dealing with atoms and molecules, the kilogram is an inconvenient unit. In this case, there is an important standard unit of mass, called the unified atomic mass unit(u), which has been established for expressing the mass of atoms as
1 unified atomic mass unit = 1u = (1/12) of the mass of an atom of carbon-12 isotope \(\left(\begin{array}{c}12\6\end{array}C\right)\) including the mass of electrons
= 1.66 × 10 –27 kg
Mass are commonly measured with common balance or beam balance, for example, in grocery stores. The masses of Planets, stars, etc., are based on the Newton's law of gravitation. . For measurement of small masses of atomic/sub-atomic particles etc., we make use of mass spectrograph in which radius of the trajectory is proportional to the mass of a charged particle moving in uniform electric and magnetic field.
Measurement of Time
A clock is used to measure the time interval of any two events. We now use anatomic standard of time, which is based on the periodic vibrations produced in a cesium atom. This is the basis of the caesium clock, sometimes called atomic clock, used in the national standards. Such standards are available in many laboratories. In the caesium atomic clock, the second is taken as the time needed for 9,192,631,770 vibrations of the radiation corresponding to the transition between the two hyperfine levels of the ground
state of caesium-133 atom. The vibrations of the caesium atom regulate the rate of this caesium atomic clock just as the vibrations of a balance wheel regulate an ordinary wristwatch or the vibrations of a small quartz crystal regulate a quartz wristwatch.

Below are the some widely used units of time:
| minute | 60 seconds |
| hour | 60 minutes, or 3,600 seconds |
| day | 24 hour, or 86,400 seconds |
| week | 7 days, or 604,800 seconds |
| month | 28-31 days, or 2,419,200 to 2,678,400 seconds |
| year | 365.25 days, or about 31,557,600 seconds |
Metric Prefixes (Table)
Table below shows the metric prefixes and their relationship to the central unit that has no prefix. Length is used as an example to demonstrate the relative size of each prefixed unit.
| Prefix | Symbol | Multiplier | Meaning |
|---|---|---|---|
| Exa | E | 1018 | 1,000,000,000,000,000,000 |
| Peta | P | 1015 | 1,000,000,000,000,000 |
| Tera | T | 1012 | 1,000,000,000,000 |
| Giga | G | 109 | 1,000,000,000 |
| Mega | M | 106 | 1,000,000 |
| Kilo | k | 103 | 1,000 |
| Hecto | h | 102 | 100 |
| Deka | da | 101 | 10 |
| Deci | d | 10-1 | 0.1 |
| Centi | c | 10-2 | 0.01 |
| Milli | m | 10-3 | 0.001 |
| Micro | μ | 10-6 | 0.000,001 |
| Nano | n | 10-9 | 0.000,000,001 |
| Pico | p | 10-12 | 0.000,000,000,001 |
| Femto | f | 10-15 | 0.000,000,000,000,001 |
| Atto | A | 10-18 | 0.000,000,000,000,000,001 |



