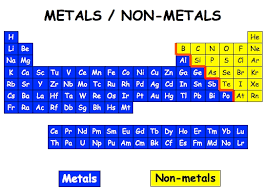
Why do metallic properties gradually change into nonmetallic in the same period?
1 Answer
On moving from left to right in the same period of modern periodic table the metallic character of elements decreases as the electropositive character of elements decreases and the electronegative character increases. So, the metallic properties gradually change into non metallic properties in the same period.
-
How would you predict the geometry of the ammonia molecule on the basis of VSEPR theory? 1
-
What kind of hybridization results in trigonal planer geometry? 1
-
Which hybridization results in a linear geometry. Give an example. 1
-
What kind of hybridization results in tetrahedral geometry? Give an example. 1
-
What is the mode of hybridization for carbon in ethyne and oxygen in water? 1
-
Predict the mode of hybridization of B in BF3. write two features. 1
-
State any two proper conditions and mode of hybridization of C in C2H2? 1