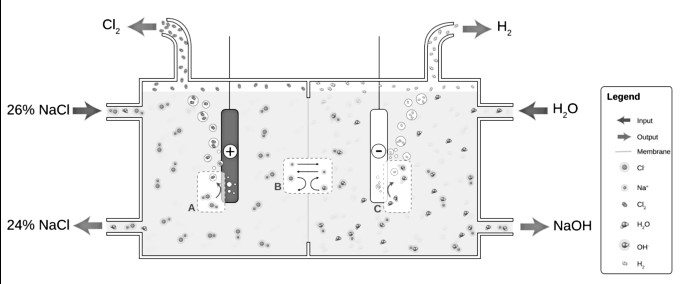
The water competes with the dissolved Na+ and Cl ions because of the oxidization and reduction property of water, as a result, hydrogen is produced instead of sodium.
The electrolysis of aqueous NaCl results in hydrogen and chlorine gas. At the anode (A), chloride (Cl-) is oxidized to chlorine. The ion-selective membrane (B) allows the counterion Na+ to freely flow across but prevents anions such as hydroxide (OH-) and chloride from diffusing across. At the cathode (C), water is reduced to hydroxide and hydrogen gas. The net process is the electrolysis of an aqueous solution of NaCl into industrially useful products sodium hydroxide (NaOH) and chlorine gas.
The reaction at Cathode is
\(H_2O(l)+2e^-\rightarrow H_2(g)+2OH^-\)
While the reaction at Anode is
\(Cl^-\rightarrow\frac12Cl_2(g)+1e^-\)
The overall reaction is as follows:
\(NaCl(aq)+H_2O(l)\rightarrow Na^+(aq)+OH^-(aq)+H_2(g)+\frac12Cl_2(g)\)
Reduction of Na+ (E° = –2.7 v) is energetically more difficult than the reduction of water (–1.23 v), so in aqueous solution, the latter will prevail.