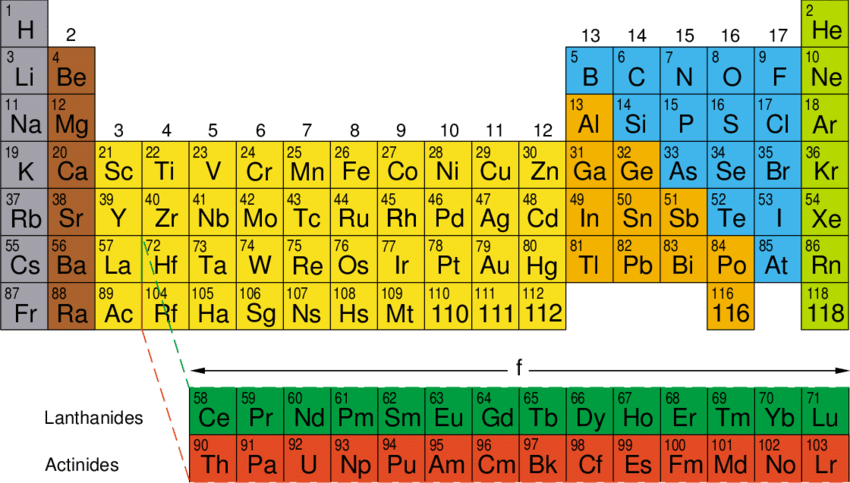
A part of periodic table is given below:
|
|
I A |
II A |
III A |
IV A |
V A |
VI A |
VII A |
O |
|
Period-2 |
Li |
Be |
B |
C |
N |
O |
F |
Ne |
|
Period-3 |
Na |
Mg |
Al |
Si |
P |
S |
Cl |
Ar |
Study the above periodic table to answer the following question.
- Write name of two most reactive non-metals.
- Name the most reactive metal.
- Draw atomic structure of that element which is placed in period -3 and has valency 4.
- Name inter gases.
1 Answer
1.
The two most reactive non metals are:
Florine (F)
Chlorine (Cl)
2.
Sodium (Na) is the most reactive metal.
3.
Silicon (Si) element is placed in period-3 and has valency -4. The atomic structure of silicon is given below:
4.
Neon (Ne) and Argon (Ar) are the inert gases.
-
How would you predict the geometry of the ammonia molecule on the basis of VSEPR theory? 1
-
What kind of hybridization results in trigonal planer geometry? 1
-
Which hybridization results in a linear geometry. Give an example. 1
-
What kind of hybridization results in tetrahedral geometry? Give an example. 1
-
What is the mode of hybridization for carbon in ethyne and oxygen in water? 1
-
Predict the mode of hybridization of B in BF3. write two features. 1
-
State any two proper conditions and mode of hybridization of C in C2H2? 1