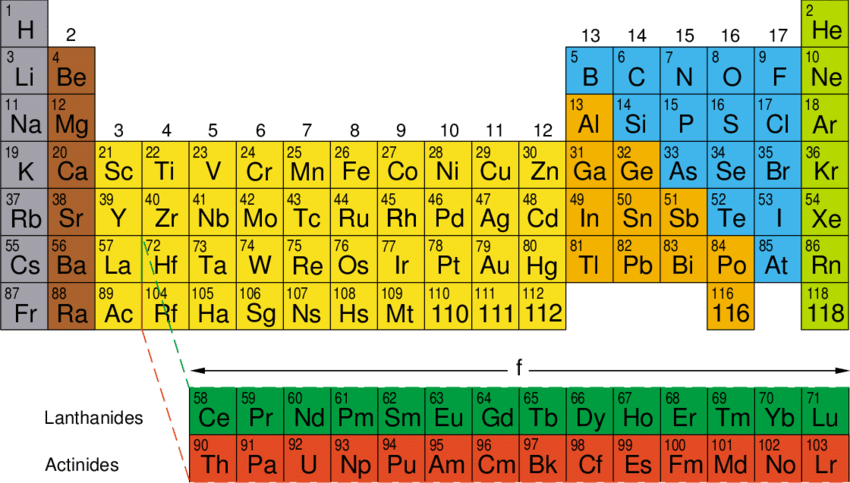
In which group and period does the element having electronic configuration 1s2 , 2s2 , 2p6 , 3s2 fall on the periodic table.
- 1st and 3
- II and 3
- 12
- III and 4
1 Answer
The correct answer is option B ( II and 3).
-
How would you predict the geometry of the ammonia molecule on the basis of VSEPR theory? 1
-
What kind of hybridization results in trigonal planer geometry? 1
-
Which hybridization results in a linear geometry. Give an example. 1
-
What kind of hybridization results in tetrahedral geometry? Give an example. 1
-
What is the mode of hybridization for carbon in ethyne and oxygen in water? 1
-
Predict the mode of hybridization of B in BF3. write two features. 1
-
State any two proper conditions and mode of hybridization of C in C2H2? 1