Introduction
The form of energy that gives the sensation of warmth is called heat. Its S.I unit is the joule (J) and the CGS unit is a calorie (Cal).
1cal=4.2J
Heat is measured by using a device called a calorie meter. Heat always flows from higher concentration to lower concentration. (From hot object to cold object)
On the basis of kinetic energy or molecular theory, Heat is defined as the total sum of the kinetic energy of the molecules of a body.
The heat contained by a body depends upon:
- Mass of the body
- Average kinetic energy of the molecules of a body.
Definition of 1 calorie heat
The amount of heat required to increase the temperature of 1 gram of pure water by 1° C or 1K is called 1 calorie heat.
Effects of heat
- It changes the volume of the body.
- It changes the physical state of a body.
- It changes the solubility of a substance.
- It brings about a chemical change.
- It changes the temperature of a body.
Temperature
The degree of hotness and coldness of a body is called temperature. Its S.I unit is Kelvin (K) whereas other units are Celsius (C) and Fahrenheit (F). Thermometer is used to measure the temperature.
On the basis of molecular theory or kinetic energy, temperature is defined as the average kinetic energy of the molecules of the body. Temperature depends upon molecular vibration of molecules.
Temperature scale and their relation
| S.N | Temperature Scale | Lower fixed point | Upper fixed point |
| 1 | Celsius | 0^\circ | 100^\circ |
| 2 | Fahrenheit | 32^{circ} | 212^\circ |
| 3 | Kelvin | 273^\circ | 373^\circ |
The relation between K, F and C is
\(\frac{C-0}{100}=\frac{F-32}{180}=\frac{K-273}{373}\)
Thermometer
The device which is used to measure the temperature of body is called thermometer. The working principle of thermometer is “liquid expands on heating and contracts on cooling.” A thermometer consists of a glass tube in which a bulb is attached with a capillary tube. The bulb is filled with thermometric liquid. When heat us supplied to a bulb the liquid in the bulb rises up in capillary tube. After some time, it shows a constant reading which gives the temperature of a body. Generally mercury or alcohol is used as thermometric liquid.
Types of thermometer
Clinical thermometer
The thermometer which is used to measure the temperature of human body is called clinical thermometer. It is also known as Doctor’s thermometer. The normal body temperature of human is 37°C or 98.6°F so the graduation in this thermometer scale ranges from 35°C to 42°C or 95°F to 108°F.
It is prismatic in structure. In this thermometer there is kink or constriction near the bulb which allows the mercury to rise in the capillary tube but does not allow the mercury to fall back into bulb after the thermometer is removed which helps to note the reading easily and accurately.

Laboratory thermometer
The thermometer which is used to measure the temperature of different objects is called laboratory thermometer. It is also known as simple thermometer. In this thermometer scale ranges from -10°C to 110°C. It is cylindrical in shape.
Maximum and Minimum thermometer
The thermometer which is used to measure maximum and minimum atmospheric temperature of 24 hours of a particular place is called maximum and minimum thermometer.
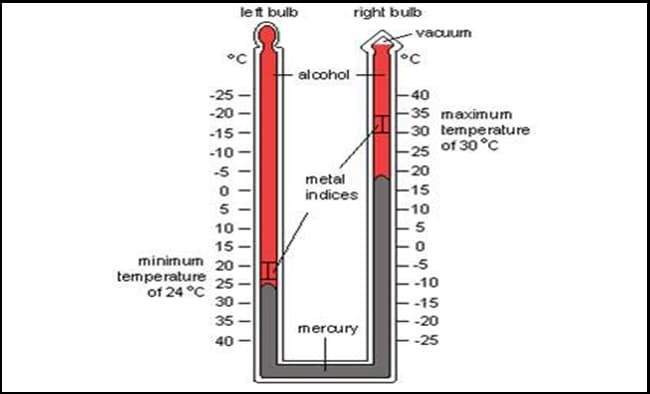
It has a U-tube which is partially filled with mercury and remaining of the tube is filled with alcohol keeping a small vacuum for expansion. The one limb is completely filled but other limb of the tube is partially filled for the vacuum. When the temperature of the surrounding increases the alcohol in left limb expands which pushes the mercury and index in the limb. When the temperature falls the alcohol in the limb contracts. As a result, the mercury flows towards the alcohol in the left limb. The movement helps to displace the index to left limb . The index of right limb gives the maximum temperature and that of left limb gives minimum temperature. (We need to note the lower point of the index). Once reading is noted, the indices are drawn to the mercury surface by using a magnet as the index is made up of magnetic substance.
Digital thermometer
The thermometer which shows the temperature in digits is called digital thermometer. A digital thermometer has a built-in-sensor which determines the amount of indicators and displays them in the monitor.
The principle of the thermometer is recording the change of electrical resistance of a conductor and converting it into a numeric value.

Heat Equation
The heat gain or lose by a body is directly proportion to mass of a body and change in temperature.
If ‘Q’ be the heat gain or lose, ‘m’ be the mass of the body and ‘dt’ be the change in temperature then,
Heat gain or lose is directly proportional to mass of body.
Heat gain or lose is directly proportional to mass of body.
Q ∝ m …………….. (i)
Heat gain or lose is directly proportional to change in temperature.
Q ∝ dt ……………... (ii)
Combining equations (i) and (ii), we get
Q ∝ mdt
Or, Q = msdt
Here‘s’ is specific heat capacity
Q = msdt
Heat equation is defined as the amount of heat gain or lost by a body is equal to product of mass of a body, change in temperature and specific heat capacity.
The heat gain of lost by a body depends on following
- Mass of the body
- Specific heat capacity
- Change in temperature.
Specific Heat Capacity
The amount of heat a require to increase the temperature of 1 kg of a substance by 1°C or 1K is called specific heat capacity. Its S.I unit is J/kg°C.
The specific heat capacity if water is 4200 J/kg°C, it means that 4200J of heat is required to increase the temperature of 1 kg of water by 1°C.
Among liquids, water has the highest specific heat capacity due to this reason water is used as a coolant to cool engines of vehicles. Also, it is used in hot water bags.
We know,
Q = msdt
If ‘Q’ and ‘m’ are constant
\(\operatorname dt\propto\frac1s\)
Change n temperature is inversely proportional to specific heat capacity. The substance having high specific heat capacity has fewer rises in temperature then the substance having less heat capacity.
Also if ‘m’ and ‘dt’ are constant, then
Q ∝ s
Heat gain or lost by a body is directly proportional to specific heat capacity. The substances having high specific heat capacity will absorb or lose more amount of heat than those having low specific heat capacity.
Calorimetry and its Principle
The measure of heat gained or lost by a body is called calorimetry.
The principle of calorimetry states that,” when a hot object and cold object are kept in thermal contact, heat lost by hot object is equal to heat gained by cold object provided that no heat escapes to the surrounding.”
Let us consider,
Mass of hot object = m1
Mass of cold object = m2
Specific heat capacity of hot object = s1
Specific heat capacity of cold object = s2
Temperature of hot object = t1
Temperature of cold object = t2
Final temperature (temperature of mixture) = t
According to principle of calorimetry,
Heat lost = heat gain
∴ m1s1(t1–t) = m2s2(t–t2)


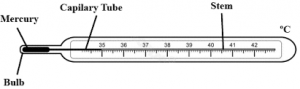 Clinical thermometer[/caption]
Clinical thermometer[/caption]

